LEQVIO is for LDL-C reduction in patients with primary hyperlipidemia along with diet and statin therapy.1

*Greater LDL-C reduction was maintained during each 6-month dosing interval vs placebo as a complement to a maximally tolerated statin.1
By adding nonstatin therapy with LEQVIO®, you can aim to achieve and remain at LDL-C target for your patients
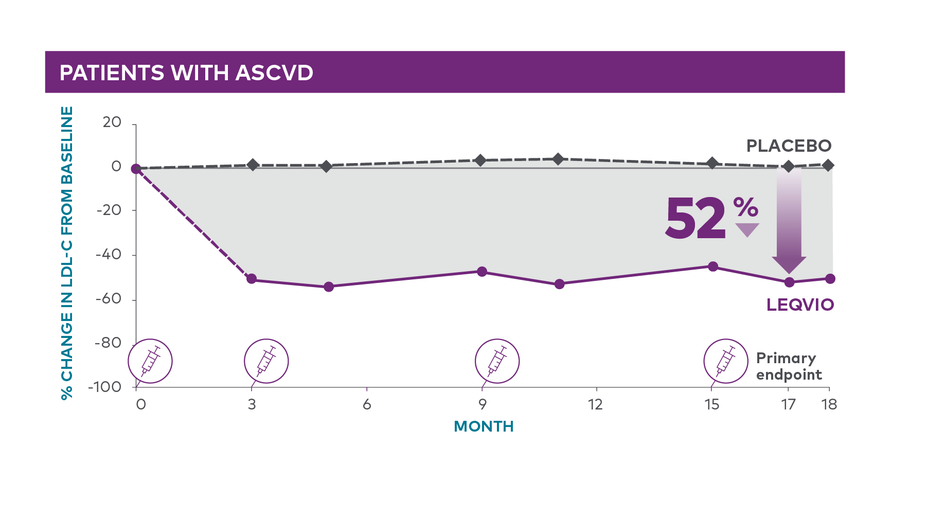
In ORION-10 (N=1561) on top of a maximally tolerated statin
Difference from placebo at month 17 (95% CI: -56%, -49%; P<0.0001)1 (LEQVIO n=781 or placebo n=780)1
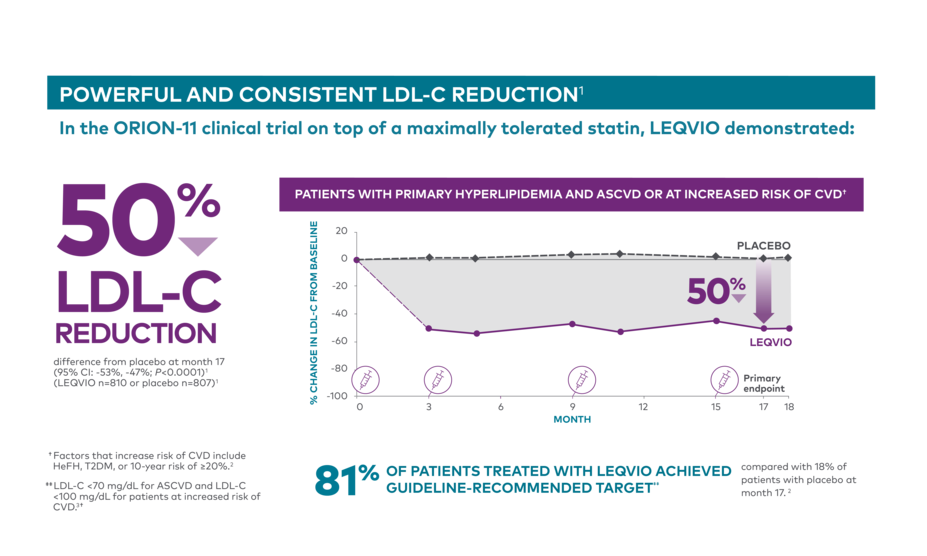
LDL-C reduction on top of a maximally tolerated statin, LEQVIO vs placebo1
In ORION-10, 84% OF PATIENTS achieved guideline-recommended LDL-C target <70 mg/dL
compared with 18% of patients on placebo at month 172
Study Design: ORION-10 (N=1561) and ORION-11 (N=1617) were multicenter, double-blind, randomized, placebo-controlled, 18-month, Phase III trials in patients with established ASCVD (ORION-10 and ORION-11) or increased risk for CVD† (ORION-11). Patients were taking a maximally tolerated statin with or without other lipid-modifying therapy and required additional LDL-C reduction. The primary efficacy measure was the percent change in LDL-C from baseline to day 510.1,3
†Factors that increase risk of CVD include HeFH, T2DM, or 10-year risk of ≥20%.2
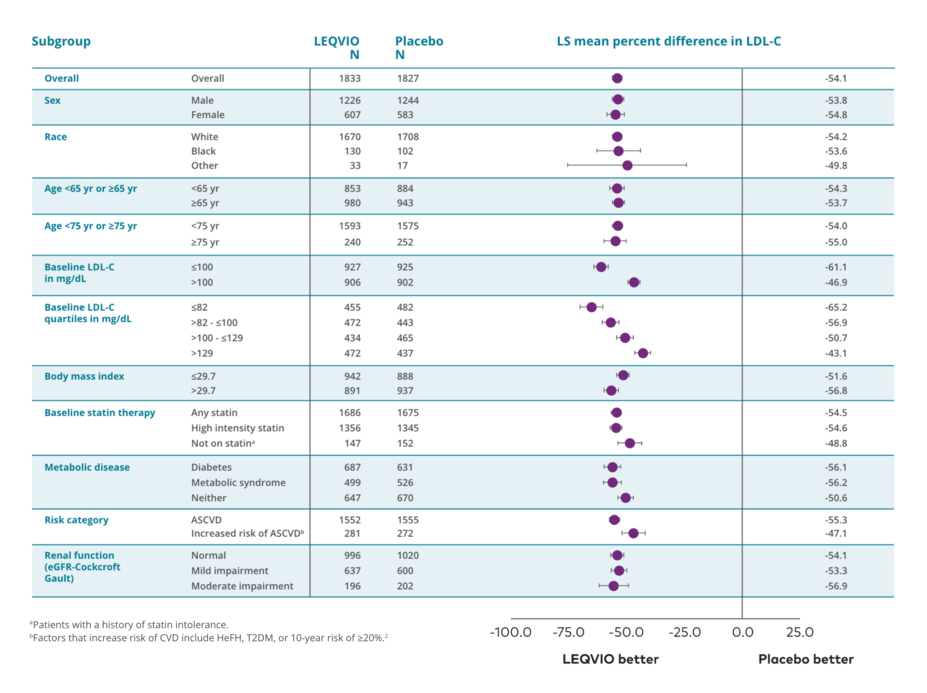
LEQVIO DEMONSTRATED CONSISTENT REDUCTION ACROSS PREDEFINED SUBGROUPS4,5
IN THE ORION-1 PHASE II STUDY, LDL-C REDUCTION WAS APPARENT WITHIN 14 DAYS AFTER THE FIRST DOSE OF LEQVIO1,6
Study Design: ORION-1 was a Phase II, multicenter, double-blinded, randomized, placebo-controlled clinical trial of 345 subjects with established ASCVD who were taking a maximally tolerated statin and required additional LDL-C reduction. Of those, 91 patients received the 284-mg dose of LEQVIO. The primary efficacy endpoint was the percent reduction in LDL-C from baseline to day 180 and was calculated for multiple time points at days 14, 30, 60, 90, 120, 150, 180, 210, and 240.1,6
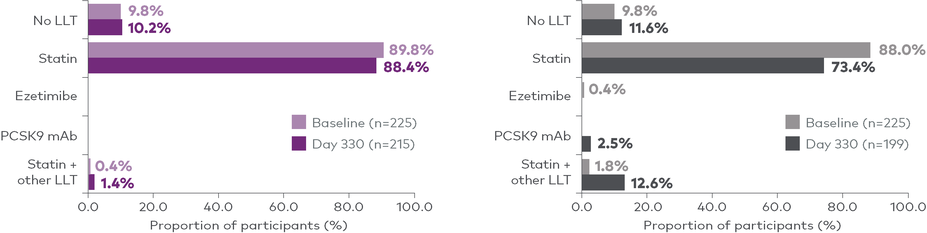
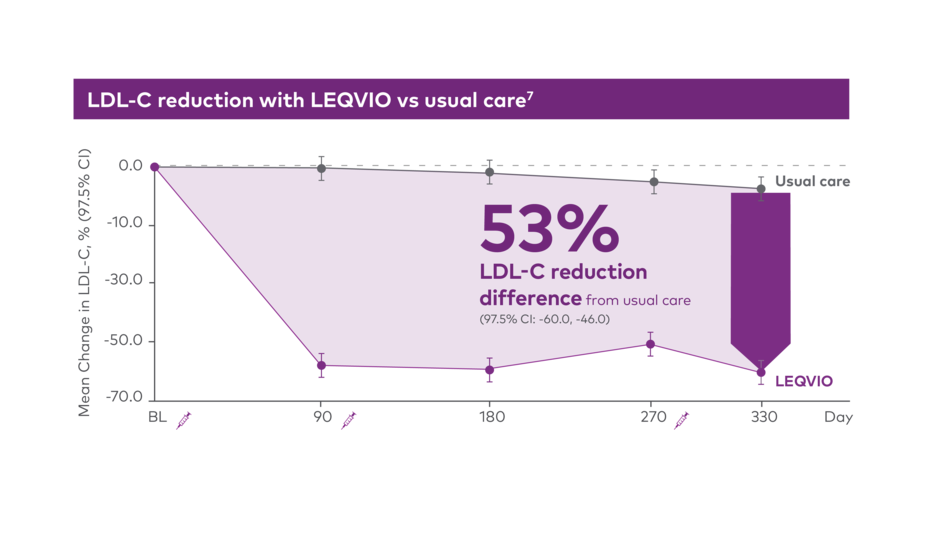
V-INITIATE compared immediate LEQVIO use after failure to reach LDL-C target‡ with maximally tolerated statin vs usual care, which was physician-determined based on the 2018 AHA/ACC guidelines.7
In the usual care arm, most patients (73%) remained on statins only, with a minority of patients receiving any additional nonstatin lipid-lowering therapy, including 10 patients who received at least 1 dose of LEQVIO.7
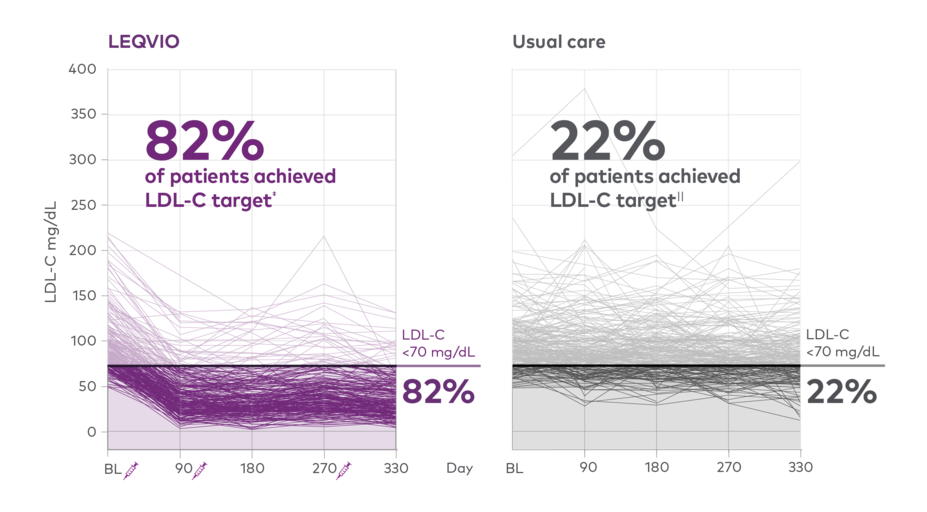
‡Guideline-recommended LDL-C target was <70 mg/dL for patients with ASCVD.2,8
Early LEQVIO use after failure to reach LDL-C target on statin7
6% of patients in the LEQVIO arm vs 17% of patients in the usual care arm discontinued statins§
[-11% (97.5% CI: -18%, -3%)]7
Study design for V-INITIATE: A 12-month, randomized, open-label, Phase IIIb study in patients with ASCVD (N=450) evaluated the efficacy and safety of early initiation with LEQVIO immediately upon failure to reach LDL-C <70 mg/dL on maximally tolerated statin vs usual care in a real-world setting.7
The co-primary end points were percentage change in LDL-C from baseline and statin discontinuation rate.7
Patients with ASCVD achieving LDL-C target7‡
||73% of patients remained on statins only at Day 330.7
Limitations: Usual care did not reflect “best practice” with little use of guideline-recommended nonstatin therapy; therefore, a comparison of efficacy or safety of LEQVIO vs other nonstatin therapies cannot be made. LEQVIO use (n=10) was permitted in the usual care arm which may impact the effectiveness of comparisons. Although the study was designed to mimic clinical practice, statin discontinuation rates may not reflect the real world due to participants’ potential behavior change as a result of being part of a study. The open-label design has the potential for bias and may present difficulties in the interpretation of results.
In ORION-8 (N=3274), an open-label extension study
~80% OF PATIENTS achieved guideline-recommended LDL-C target at end of study#**
(78%; 95% CI: 77, 80)2,8,9
~50% LDL-C REDUCTION at EOS
n=2731 (mean percentage change in LDL-C was -49% at EOS**; 95% CI: -50%, -48%)9
Study Design: ORION-8, an open-label extension trial that included 3274 patients from ORION-9, ORION-10, ORION-11, and ORION-3, was designed to assess the long-term safety, efficacy, and tolerability of LEQVIO in patients with ASCVD or at increased risk for CVD† and elevated LDL-C, despite ongoing treatment with lipid-lowering therapy.3,9
Limitations: Study was not blinded nor controlled and includes inherent self-selection bias for continuing onto the extension trial. The open-label design and absence of a control group may present difficulties in the interpretation of results, allowing comparisons only to baseline values.
†Factors that increase risk of CVD include HeFH, T2DM, or 10-year risk of ≥20%.2
¶209 (6.4%) patients had exposure to LEQVIO for 6+ years, 213 (6.5%) patients had exposure to LEQVIO for 5+ years, 1553 (47.4%) patients had exposure for 4+ years, and 2095 (64%) patients had exposure for 3+ years.10
#LDL-C target was <70 mg/dL for patients with ASCVD and <100 mg/dL for patients at increased risk for CVD§ and aligns with AHA/ACC guidelines.8,9
**End of study was defined as day 1080 or ≥90 days after the last LEQVIO dose.9
THERE’S MORE TO KNOW ABOUT LEQVIO
Discover what makes LEQVIO dosing different