
LEQVIO® IS A FIRST-IN-CLASS siRNA LIPID-LOWERING THERAPY THAT TARGETS THE LIVER1
LEQVIO prevents formation of PCSK9 protein, allowing more LDL-C receptors to remove circulating LDL-C1

Works selectively in the liver1,2
- Harnesses the body’s process of RNA interference, which is restricted to the cytoplasm2,3
- No impact on DNA2,3

Reaches undetectable levels in circulation within 48 hours of administration1
- Half-life of 9 hours; no accumulation with multiple dosing1
Prevents the production of PCSK9 protein with an extended duration of
effect1-3
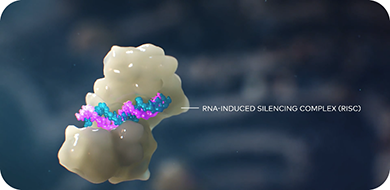
- Is slowly released into the cytoplasm and loads onto the RNA-induced silencing complex (RISC)2,3
- Once loaded, works with RISC to sequentially cleave multiple copies of PCSK9 protein mRNA, which continuously prevents PCSK9 production1,2
- Less PCSK9 allows for increased LDL-C receptors to bind to and decrease circulating LDL-C1
Explore the MOA
Discover more about how LEQVIO works differently than other LDL-C–lowering treatments.

Dive into siRNA technology
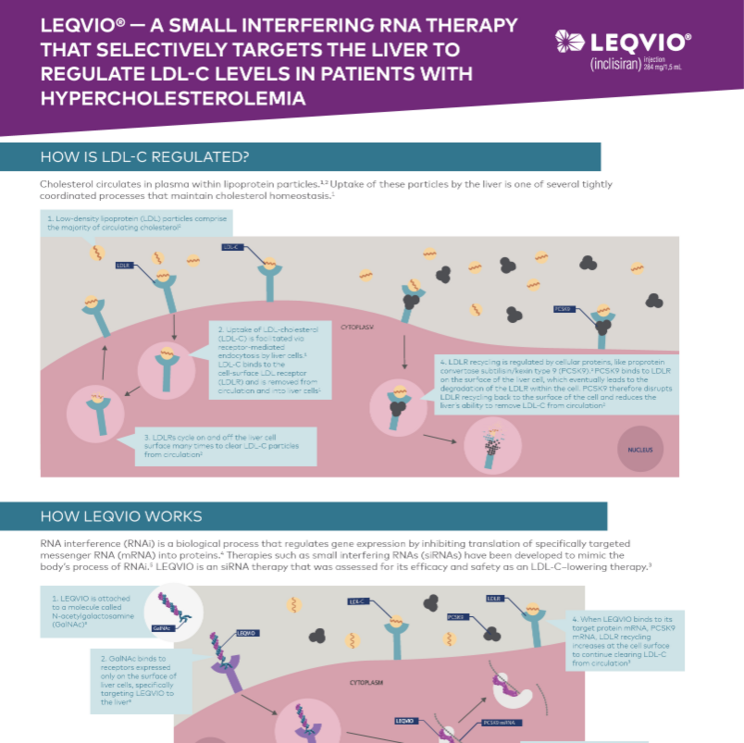
Read the infographic to learn more about LEQVIO—a small interfering RNA therapy that selectively targets the liver to regulate LDL-C levels.
There's more to know about LEQVIO
See essential dosing and administration information for LEQVIO
DNA, deoxyribonucleic acid; LDL-C, low-density lipoprotein cholesterol; MOA, mechanism of action; mRNA, messenger ribonucleic acid; PCSK9, proprotein convertase subtilisin/kexin type 9; RNA, ribonucleic acid; siRNA, small interfering ribonucleic acid.